UE Systems introduces UE ONE connected platform
UE Systems today announced UE ONE™, a connected platform designed to change the way industry listens to its equipment, turning what it hears into diagnostic evidence, clearer decisions, and meaningful action.
As organizations expand condition monitoring programs and face increasing pressure to improve uptime, reduce costs, and operate more efficiently, many still struggle to connect data collection with action. UE ONE addresses this challenge by linking ultrasound data, analysis, and operational workflows into a unified environment that helps teams detect earlier, diagnose faster, and act with confidence.
“Customers don’t need more data. They need clarity,” said Blair Fraser, Executive Vice President, Global Technology & Business Development at UE Systems. “UE Systems provides the products. UE ONE connects the workflows. Reliability teams scale the value.”
UE ONE represents a shift from isolated inspections and disconnected tools toward a more connected, insight-driven approach to reliability. The platform brings together diagnostic ultrasound data, analysis, interpretation, alerts, and reporting, helping teams move from identifying change to understanding what the signal is showing and what action should follow.
At its core, UE ONE is built on a simple idea: ultrasound that listens, paired with intelligence that acts. The platform transforms raw signals into diagnostic evidence and supports users in interpreting that evidence with greater clarity and consistency. Advanced analysis capabilities help teams move beyond simple level readings, while UE ONE | Copilot assists in translating findings into clear, actionable insights that can be shared and acted on across teams.
Rather than replacing expertise, UE ONE is designed to make it easier to apply and scale. It helps organizations reduce reliance on individual specialists, improve consistency across sites, and make ultrasound knowledge more repeatable.
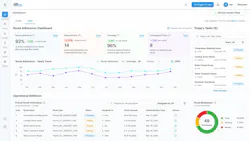
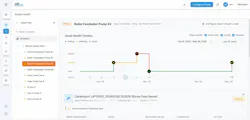
Beyond analysis, UE ONE connects the workflows that surround ultrasound programs. Inspection routes, analysis processes, alerts, dashboards, reporting, and administration are integrated into a single environment, enabling organizations to standardize how work is executed, reviewed, and communicated. This allows teams to move from isolated inspection activity to scalable, program-level visibility.
The platform is designed to work across environments and systems, supporting data from anywhere and providing a platform for anyone looking to scale ultrasound-driven reliability. With flexible deployment options across cloud, customer-managed, and on-premises environments, UE ONE™ can adapt to a wide range of operational and regulatory requirements.
“Ultrasound has always been a powerful way to detect problems early,” Fraser added. “UE ONE builds on that foundation by helping teams connect what they hear to what they do next.”
UE ONE is being showcased at Ultrasound World, where attendees have the opportunity to experience the platform firsthand.